 A chemist has constructed a galvanic cell consisting of two beakers. An electrolytic cell consumes electrical energy from an external source to drive a nonspontaneous chemical reaction. #2("Ag"^(+)(aq) + cancel(e^(-)) -> "Ag"(s))# A galvanic (voltaic) cell converts the energy released by a spontaneous chemical reaction to electrical energy. Recall that oxidation takes place at the anode and reduction takes place at the cathode. The reaction conditions ( pressure, temperature, concentration, etc.), the anode, the cathode, and the electrode components are all described in this unique shorthand. Thus, we reverse the other half-reaction to write it as an oxidation and the decomposition of sulfurous acid leads to the oxidation of sulfur dioxide to sulfate at the anode. Cell notations are a shorthand description of voltaic or galvanic (spontaneous) cells. Anode half-reaction: + 2OH + 2e Cathode half-reaction: Ni Cu + 20 + 2 OH Shorthand notation: Cu (OH)2 () - Ni (OH)2 (8) Answer Bank KOH (aq) Ni (s) KOH (aq) NI. 
The electrode on the left is the anode, and the one on the right is the cathode. The silver reduction has a more positive so reduction is more spontaneous for #"Ag"^+# and #"Ag"^(+)# is reduced at the cathode. Complete the half-reactions for the cell shown, and show the correct shorthand notation for the cell. #"SO"_4^(2-)(aq) + 4"H"^(+)(aq) + 2e^(-) -> "SO"_2(g) + 2"H"_2"O"(l)#, = +"0.20 V"# For the galvanic cell reaction, expressed below using shorthand notation, what half-reaction occurs at the anode Not the exact question youre looking for Post any question and get expert help quickly. cathode and aluminum is at anode is: (Consider the given half equations): Cu2+. #"SO"_4^(2-)(aq) -> "H"_2"SO"_3(aq) -> "H"_2"O"(l) + "SO"_2(g)#Īs a result, we find the two half-reactions to be: According to the following shorthand notation of a voltaic cell, the anode is. 
cathode, in the cell notation used above the cathode is on the.  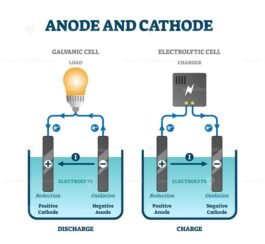
Ignoring the charge balance, the main action going on in one half-reaction is: In an electrolytic cell, the negative terminal is the (cathode/anode) and is the site of the (oxidation/reduction) half-reaction. Anode (oxidation occurs at the anode, in the cell notation used above the anode is on the. anode (oxidation): Zn(s) Zn2+(aq) + 2e cathode (reduction): Cu2+(aq) + 2e Cu(s) overall: Zn(s) +Cu2+(aq) Zn2+(aq) + Cu(s) (17.2.13) (17.2.14) (17.2.15) Zn(s)Zn2+(aq)Cu2+(aq)Cu(s). In the SI system of units, the value of the elementary charge is exactly defined as e īy combining the best measured value of the antiproton charge (below) with the low limit placed on antihydrogen's net charge by the ALPHA Collaboration at CERN.There's a small trick here. The elementary charge, usually denoted by e, is a fundamental physical constant, defined as the electric charge carried by a single proton or, equivalently, the magnitude of the negative electric charge carried by a single electron, which has charge −1 e. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
